SUGAR Study
An Audit of Concordance with ANZCA Perioperative Diabetes Management Guidelines
ACROSS AUS + NZ HOSPITALS | MULTIPLE SPECIALTIES | TWO-WEEK DATA COLLECTION PERIODS
The study is planned to commence mid-2026
Expression of Interest Form: SUGAR EOI Form
What is the SUGAR Study?
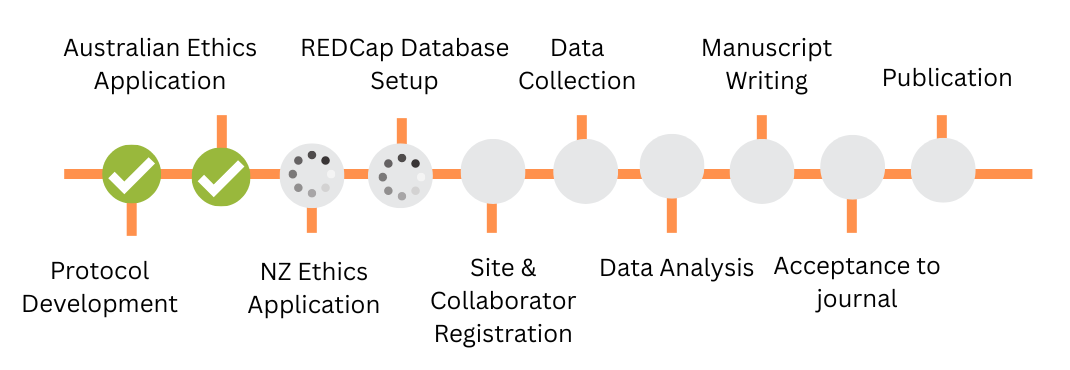
Project Status
What Are Our Main Objectives?
SUGAR is an Australian and Aotearoa New Zealand, prospective multi-centre cohort study that aims:
- Audit compliance with perioperative diabetes guidelines for patients undergoing elective surgery in Australia and Aotearoa New Zealand
- Explore associations between concordance with guideline recommendations and incidence of postoperative complications
Through the SUGAR Study, we aim to continue building collaborative research capacity within medical students and junior doctors in ANZ.
Who Will Be Involved?
TASMAN invites all medical students in their clinical years, junior doctors and consultants to become collaborators of this study.
All collaborators will be included as PubMed-citable co-authors on resulting publications. All hospitals conducting common elective surgical procedures can participate in this study. Full list of eligible procedures can be found in the protocol. This study is across Australia and Aotearoa New Zealand.
All collaborators will be included as PubMed-citable co-authors on resulting publications. All hospitals conducting common elective surgical procedures can participate in this study. Full list of eligible procedures can be found in the protocol. This study is across Australia and Aotearoa New Zealand.
-
- If you are in Australia or New Zealand, please fill out an EOI through this link: SUGAR EOI Form
- If you have any further queries please email us at tasmananz@gmail.com.